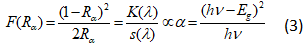HTML / Full Text View Download Manuscript
Nano Progress
Research Article
Optical, Electrical and Magnetic Properties of Dy-Substitution on BiFeO3 Nanoparticles
Abstract: [Bi1-xDyxFeO3, x=0.05 (BDF1), Bi1-xDyxFeO3, x=0.10 (BDF2), and Bi1-xDyxFeO3, x=0.15 (BDF3)] nanoparticles have been successfully synthesized by co-precipitation method. From X-ray diffraction data analysis confirms the phase formation and structural transformation from rhombohedral structure to orthorhombic when increase in the Dy concentration into BFO with evidence of Rietveld refinement analysis. The average crystallite sizes were calculated from X-ray diffraction analysis for BDF1, BDF2 and BDF3 were found to be 48, 33, 28 and 26 nm respectively. The band gap values of the BDF1, BDF2 and BDF3 samples using Kubelka– Munk (K-M) function evaluated from the diffuse reflectance spectra studies values are 2.27, 2.57 and 2.59 eV, respectively. Differential Thermal Analysis (DTA) shows the existence of two prominent peaks related to ferroelectric ordering and melting point of Dy substituted BFO. From the magnetic measurement for BDF1 showed antiferromagnetic behaviour and for BDF2 and BDF3 samples shows weak ferromagnetic behaviour. Further, we have demonstrated the magneto electric effect for the all the samples.
Keywords: BiFeO3; Co-Precipitation; Optical; Magnetic Materials
Publication details: Received: 25th February 2019; Revised: 10th March 2019; Accepted: 18th March 2019; Published: 18th March 2019
1. Introduction
Recent years, Bismuth ferrite oxide materials have been widely studied related to magneto electric effect and these materials are classified from multiferroic materials. It has varies applications in an information storage, spintronic devices, optical devices and sensors.[1,2] Bismuth ferrite has been exhibited multiferroic materials at room temperature as it has ferroelectric ordering temperature (TC ~1103 K) and magnetic ordering temperature (TN ∼ 643 K).[3] BFO has both ferroelectric and magnetic properties and the ferroelectric property originates from the Bi–O hybridization due to the stereo-chemical activity of lone pair electrons (Bi 6s2) and the origin of magnetism arises from partially filled “d” shells of F3+ ions, which is two ordering parameters are mutually exclusive in principle.[4,5] In bulk BFO has antiferromagnetic behavior, which can explains due to the arrangement of spiral spin structure of crystals with an incommensurate long wavelength period of 62 nm. However, it has some major drawbacks are there such as presence of secondary phase along with BFO, small remnant polarization, high leakage current, low magnetization, high coercive field etc. so that BFO is difficult to use in practical applications. These drawbacks can be overcome by different modification in the synthesis technique of BFO,[6] suitable dopants at either A-site[7–9] or B-site[10–12] and co-doping at both A and B-sites.[13-15] However, there are some drawbacks in few methods which have, for example, the solid-state reaction method requires for high temperature heat treatment to formation of the single-phase BFO ceramics. For hydrothermal method is taking longtime process for formation BFO and also some difficulties to get uniform nano-sized BFO particles by other chemical methods. Moreover, there are very few reports in the literature on optical properties of BFO nanoparticles. Although some reports on the optical band gap of pure and rare earth modified BFO with different methods have been reported, such as the optical band gap of BFO 2.82 eV by wet chemical method,[16] solid state reaction (2.25 eV),[17] Sol-gel (2.66 eV),[18] magnetron sputtering 2.80 eV.[19] In addition to the potential magnetoelectric applications, BFO might find application as photocatalytic material due to its small band gap.[20] Choi et al. on the BFO photovoltaic properties has created a lot of attention in its optoelectronic applications.[21] which is a low temperature process with much less cost, can also produce pure phase having awide range of optical gap, provided a decent route and suitable precursors are employed. In addition, to improve the multiferroic properties of BFO, partial doping of rare earth ions such as La3+, Ce3+, Pr3+, Nd3+, Sm3+, Dy3+, Ho3+, Er3+ at Bi-site often demonstrated for developing its functionalities. The reasons for Dy3+ ions doping into the BFO are: (i) to suppress the impurity phases of BFO.[17] (ii) to reduce leakage current density to obtain improved ferroelectric properties.[22] and (iii) to enhancing magnetization by modifying the spiral spin structure, which may be due to of the large magnetic moment of Dy3+ (∼10.6 μB) ions.[23,24] To expected structural transition from rhombohedral to other structure for higher concentration of Dy3+ ions content in BFO, because of the Dy3+ (0.912Å ) is smaller than that of Bi3+ (1.03Å).[25,26] Xu et al. reported that enhanced ferroelectric properties by substitution of Dy ions in to BFO samples.[27] Both ferroelectric and piezoelectric properties has been improved by substitution of higher concentration of Dy ions into BFO, reported by Sun et al.,[28] Our previous work, we have reported that structural phase transformation rhombohedral to orthorombic struture for higher concentration of Dy3+ ions substituted into BFO by X-ray diffraction analysis at the same time explains about the changes from ferroelectric to paraelactric behavior for BDF3 shows a transformation from non-centrosymmetric (R3c) to centrosymmetric (Pnma) system by P-E hysteresis loop studies. In this paper, we report the influence of Dy substituted on BFO optical, electrical and magnetic properties of BFO nanoparticles.
2. Experimental Section
The synthesis procedure was published elsewhere.[29] The phase identification was examined on a Rigaku (D/Max ultima III) X-ray diffractometer using CuKα radiation. XRD data was collected at slow scan rate of 0.05°/min and simulation of crystal structure was done based on the measured XRD data and Rietveld crystal structure refinement software General Structure Analysis System (GSAS). Differential Thermal Analysis (DTA) was using the SII Nanotechnology Inc., Japan. UV- Visible studies by JASCO (Modal V-670, USA) and Luminescence studies taken by Fluorescence Spectrometer (FP-8500 Japan). The magnetic measurements were done with a vibrating sample magnetometer (Lakeshore, USA 7404).

3. Results and Discussions
3.1. Structural Studies
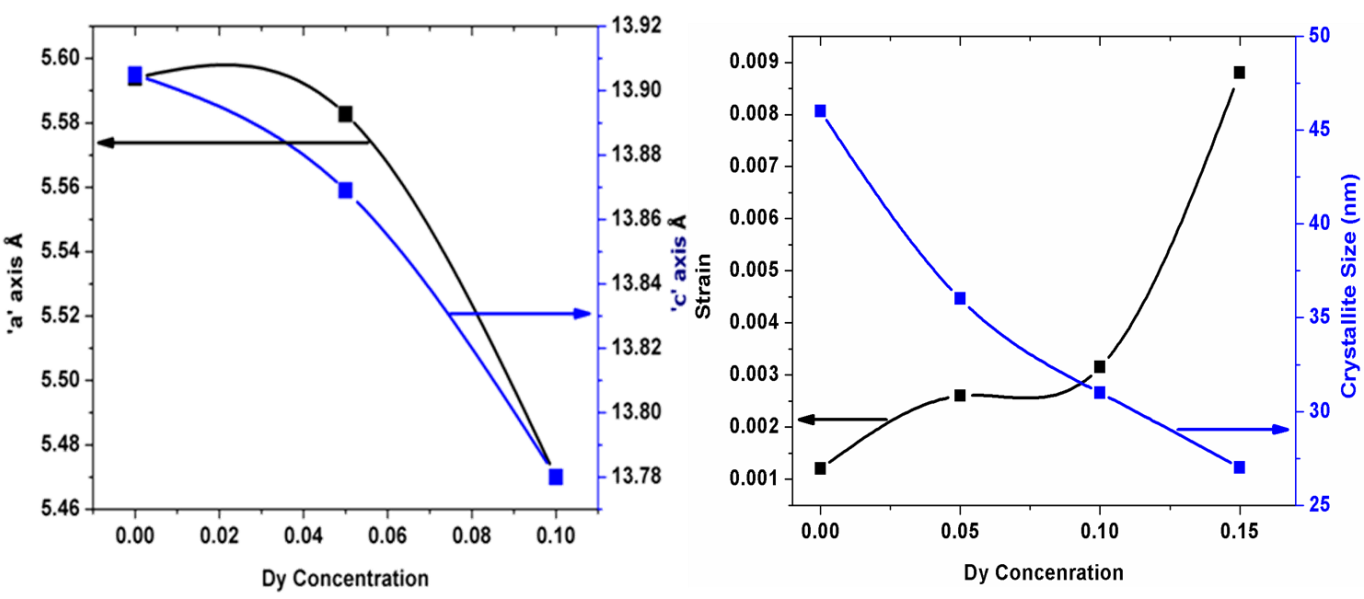
X-ray diffraction patterns of BDF1, BDF2 and BDF3 powders annealed at 600°C for 2 hrs. It has been observed the XRD patterns can be indexed to rhombohedral perovskite structure similar to the pure BFO published elsewhere.[30] Further, know the crystallography information for the prepared the samples is performed by Reitveld refinement. It has been determine the crystal structure of BDF2 and BDF3 samples shows rhombohedral structure with space group of R3c similar to pure BFO.[29] However, XRD pattern of BDF3 shown to be phase pure and has an ortho-rhombic DyFeO3- like profile (space group Pnma).[31,32] The lattice parameters and volume values are changes to the substitution of Dy concentration which are shows in Fig1(a). It has been suggest that Rp, wRp and χ2 values are good agreement with the simulated XRD patterns and the experimental data. It has suggested that structural changes from rhombohedral to orthorombic crystal structure, while increasing Dy concentration into BFO.
The average crystallite size can be calculated from X-ray diffraction data to evaluate the peak broadening of the BFO lattice. However, the full width half maximum (FWHM) peak is not only due to the particle size it is depends the strain on the instrumental peak broadening. So we can consider both the breadth of the measured peak broadening to the sample and instrumental. Since, it is require to collect a diffraction pattern from standard material as silicon to determine the FWHM of instrumental broadening.[33]
The instrument corrected broadening βhkl corresponding to the chosen diffraction peak has been estimated by using equation (1).
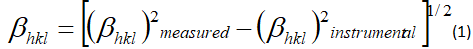
The average crystalline size and strain can be calculated from Williamson and Hall (W-H) method, equation given by
The induced strain broadening is given by ε= β hkl/ tanθ, βhkl is the full width at half maximum of the corresponding hkl plane, shape factor of the sample is ‘k’, ‘λ’ is the X-ray wavelength, ‘θ’ is the Bragg angle, ‘D’ is the average crystallite size.
W-H plotted as x-axis is 4 sinθ and with respect to βh k l cosθ along the y-axis. From the plot, we calculated the lattice strain and average crystallite size from the linear fit to the data, the value of the strain is evaluated from the slope of the line and determined crystallite size resultant from the intersection of linear line with the vertical axis both values are shows in Fig 1(b).
3.2. TEM Analysis
The images of BDF1, BDF2 and BDF3 nanoparticles show in Fig (2). All the images are measured in the range at 50nm. The average particle size is calculated from Image J software and x=0.05 sample shows around 50–70 nm, and spherical shaped nanoparticle. For x=0.10 and x=0.15 samples could not calculated the average particle size because of agglomerations in the samples.

3.3. FTIR Analysis
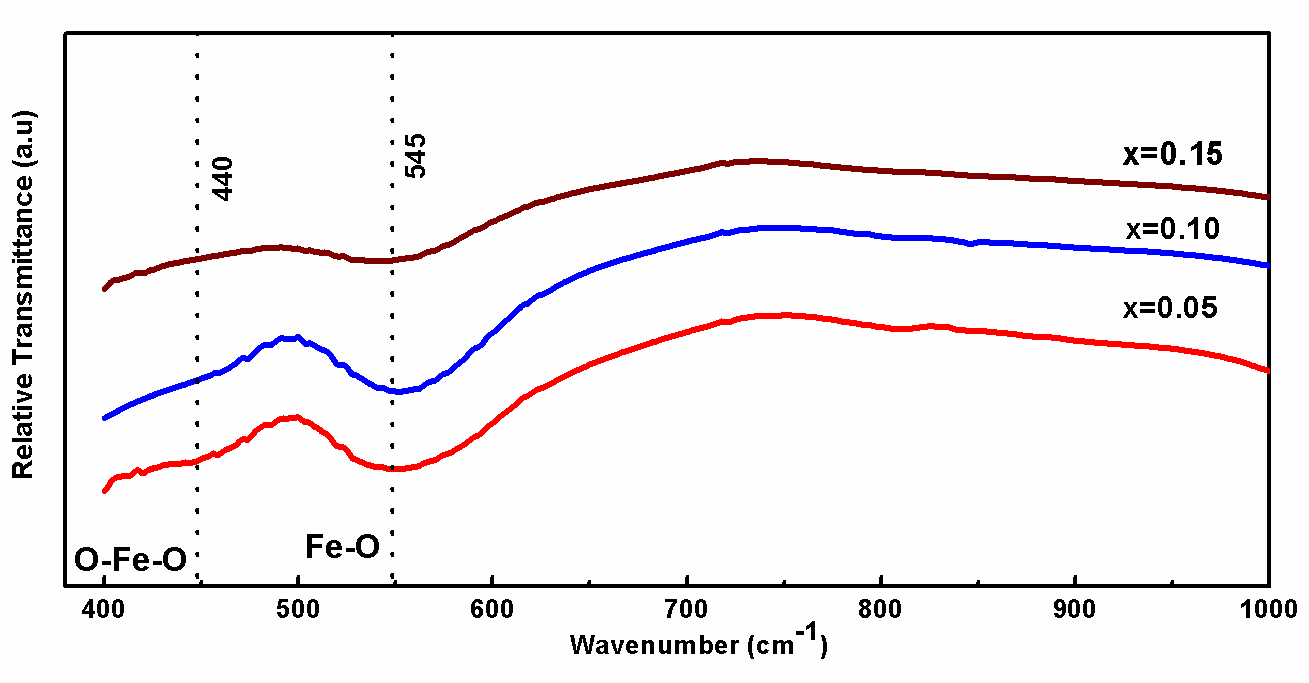
Fig. 3. Shows the BDF1, BDF2 and BDF3 nanoparticles observed bands between 700 and 400 cm-1 were mainly attributed to the formation of metal oxides. Specifically, two strong absorptive peaks to confirm the metal oxide compound around 545 and 440 cm-1 are attributed to the Fe-O stretching and O-Fe-O bending vibrations, being characteristics of the octahedral FeO6 groups in the perovskite compounds. The formation of perovskite structure can be confirmed by the presence of metal-oxygen band.[34]
.png)
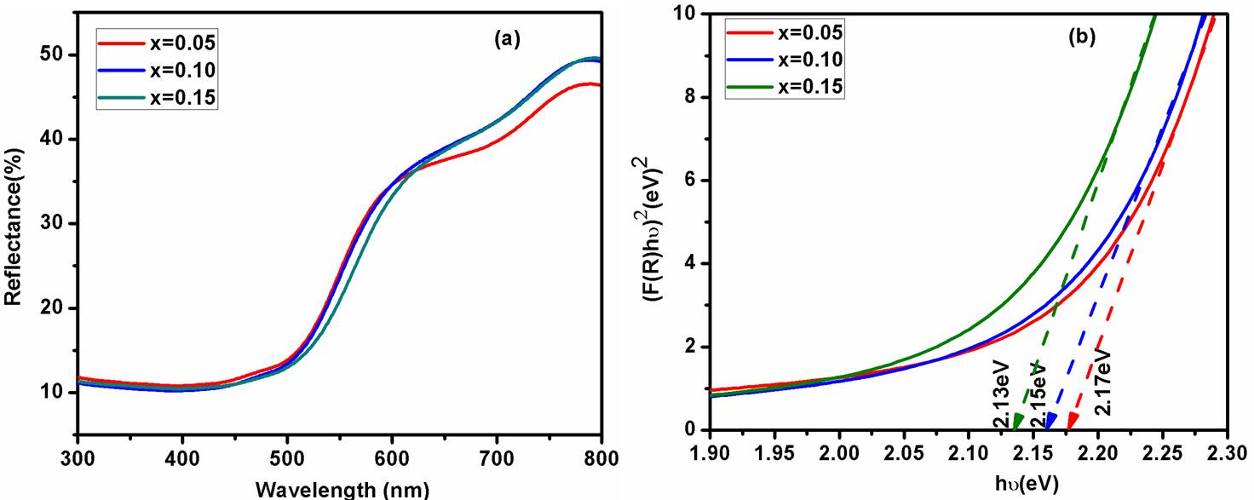
3.4. UV–Visible Diffuse Reflectance Spectra
BDF1, BDF2 and BDF3 nanoparticles are measured UV–Vis diffuse reflectance spectra (DRS) as shown in Fig. 4(a). At 500 nm, a decrease in reflectance is seen due to fundamental absorption between valance bands to conduction band by the BFO. Further, the optical band gap is calculated using K–M function[35] equation is given by:
Here, ‘F(Rα)’ is the K–M function, ‘Rα’ is the diffuse reflectance of an infinitely thick sample, ‘K(λ)’ is the absorption coefficient, ‘s(λ)’ is the scattering coefficient, ‘hν’ is the photon energy and ‘Eg’ is the band gap energy for direct transition. In present work, the direct optical band gap energy is evaluated by K-M function and the graph plotted between (F(R)hν)2 with respect to (hν) shown as an in Figure 4(b). The band gap energy is found to be around 2.18, 2.16 and 2.13 eV around 2.18, 2.16 and 2.13 eV for BDF1, BDF2 and BDF3 samples respectively. It has been observed the changes of in band gap energy of Dy doped BFO nanoparticles shows the incorporation of the Dy ions into BFO lattice. However, the band gap energy is decreases with increasing of Dy concentration, which may be due to the defect levels introduced between valance band and conduction band.
3.5. Photoluminescence Spectra
Figure 4 shows the photoluminescence properties exhibited by the prepared BDF1, BDF2 and BDF3 nanoparticles under 365 nm excitation and the emission was recorded at room temperature in the wavelength range of 350–700 nm. The enhancement of luminescence emission peaks for higher concentration of BDF3 due to the Dy3+ ions incorporation of Bi3+. Four emission peaks positioned at 411 (3.01 eV), 437 (2.83 eV), 468 (2.64 eV) and 539 nm (2.30 eV) have been observed in photoluminescence spectrum shows in fig. 5. The emission peak centred at 468 nm is to defect related emission, due to the presence of defect trap states, below the conduction band which are ascribed to defects in the grain boundaries.[36] Peak at 411 (2.99 eV) and 539 nm (2.30 eV) is observed which is attributed to the oxygen vacancy defects.[37] Absorption bands at 437 (2.83 eV) attributed to the 6A1 (6S) → 4T2(4D) transition.[10] These luminescent emissions open up many opportunities of nanocrystalline of Dy substituted in BFO samples for both further fundamental studies and nanoscale optical applications.[38]
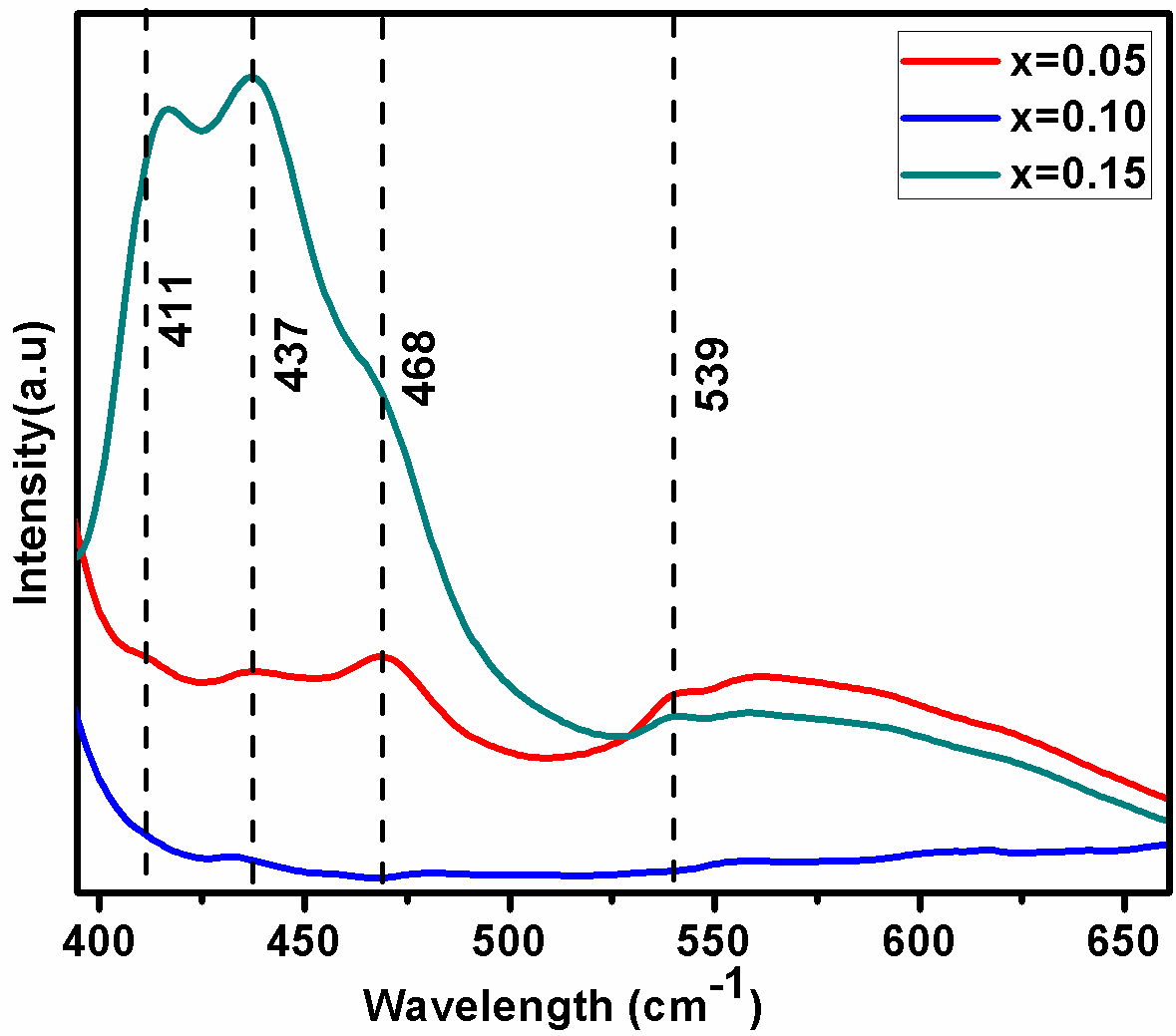

Fig. 5. shows room temperature photoluminescence emission spectrum of the synthesized BDF1, BDF2 and BDF3 nanoparticles observed at excitation wavelength 365 nm.
3.6. Thermal Analysis
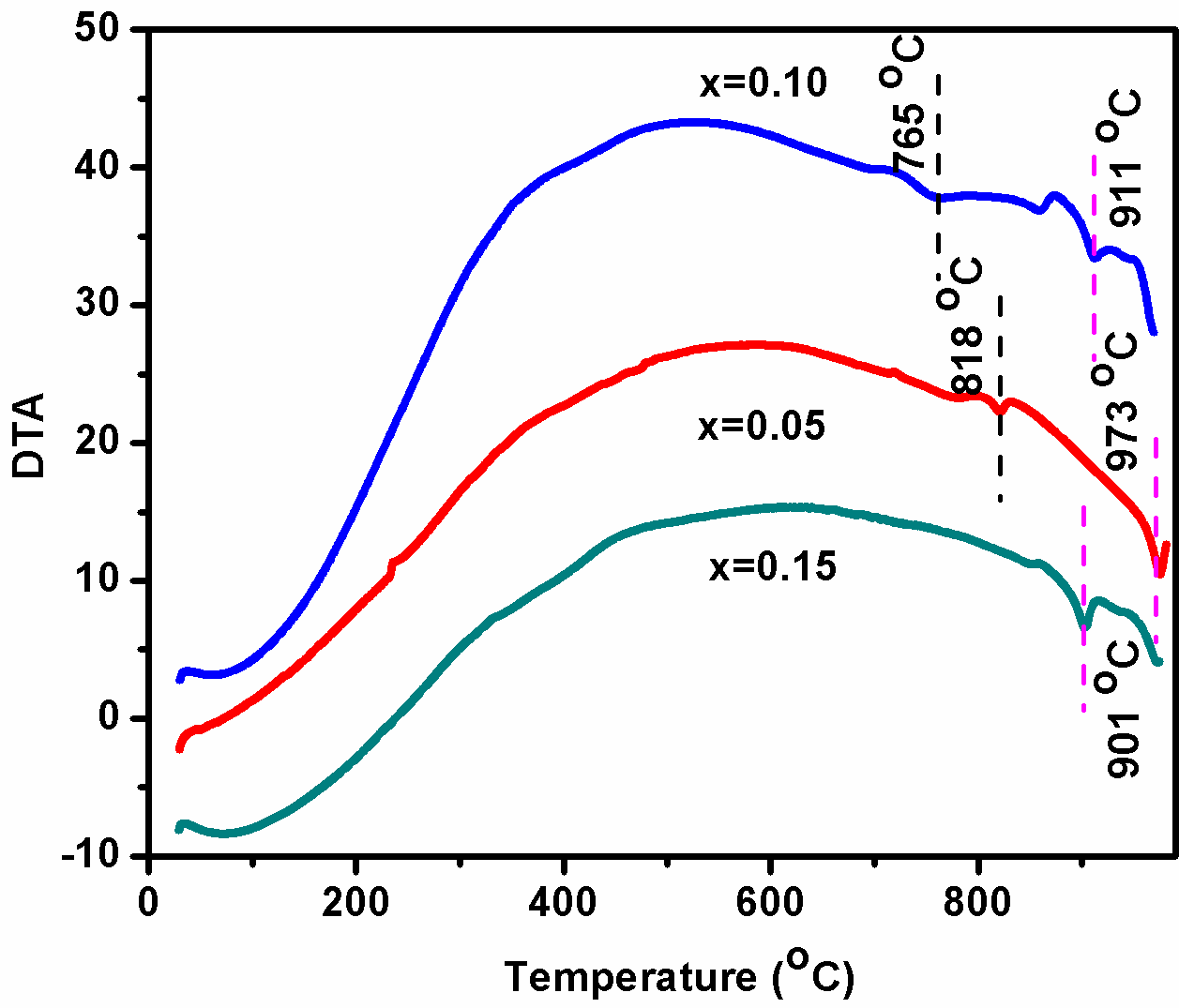
BDF1, BDF2 and BDF3 nanoparticles are measured Differential Thermal Analysis (DTA) for heating rate at 10 â—¦C/min as shown in Fig. 6. It has been observed two small peaks have been observed around 818, 765 ºC for BDF2 and BDF3respectively, these peaks are corresponds to the ferroelectric to paraelectric transition temperature.[39] The DTA measurement results show that the Curie temperature (Tc) decreases with the increase in Dy concentration. Moreover, another peak were observed around 973, 911 and 901â—¦C corresponds to the heat loss at the melting points of BDF1, BDF2 and BDF3 ceramics. These observations suggest that the Dy substitution lowered the ferroelectric phase transition and also decreases the melting point temperature.
3.7. Leakage Current Density Measurement
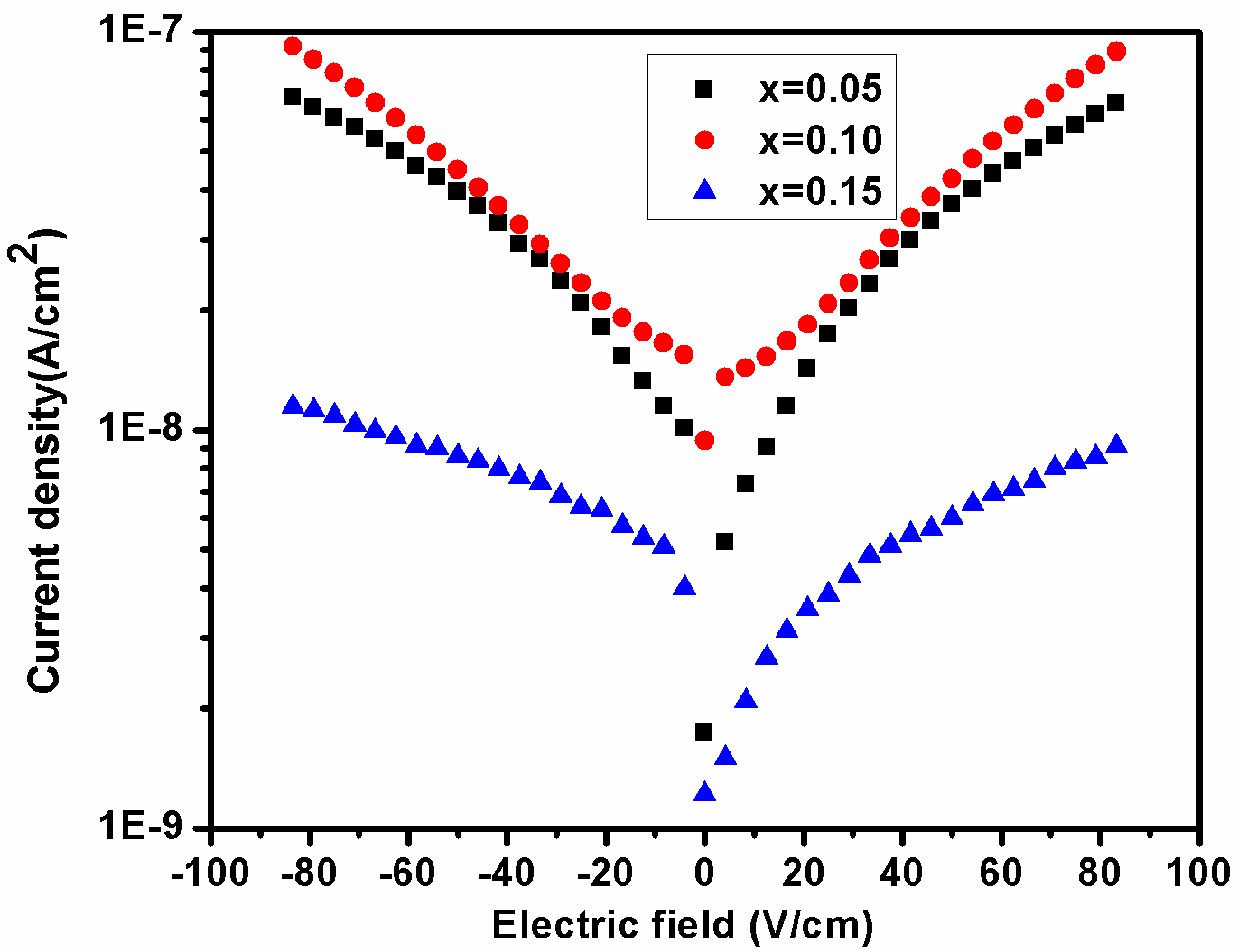
To investigate the leakage current density behavior of the BDF1, BDF2 and BDF3 samples, the leakage current were measured both the forward and reverse bias conditions attributed to the bulk-limited conductions in Dy doped BFO as shows in fig. 7. It is noticed that the effects of Dy concentration increases with decreasing the leakage current density of the BFO. An obtained the leakage current density values are 10−7 A/cm2 at 90 V/cm for BDF3, these values are smaller than that of BDF1. However, several reasons are contributing to the leakage current mechanism in rare earth metal ions substituted into BFO samples have been reported, like: (1) The leakage current in BFO is usually attributed to the space charges (such as oxygen vacancies) induced mainly by Bi volatilization. (2) Oxygen vacancies are created by substitution of rare earth metal ions into BFO (3) Fe2+ and Fe3+ are plays an important role in electronic conduction.[40] In concluded the results, it can be effectively reduced by the substitution of rare earth elements into BFO.[41,42]
3.8. Magnetic Properties
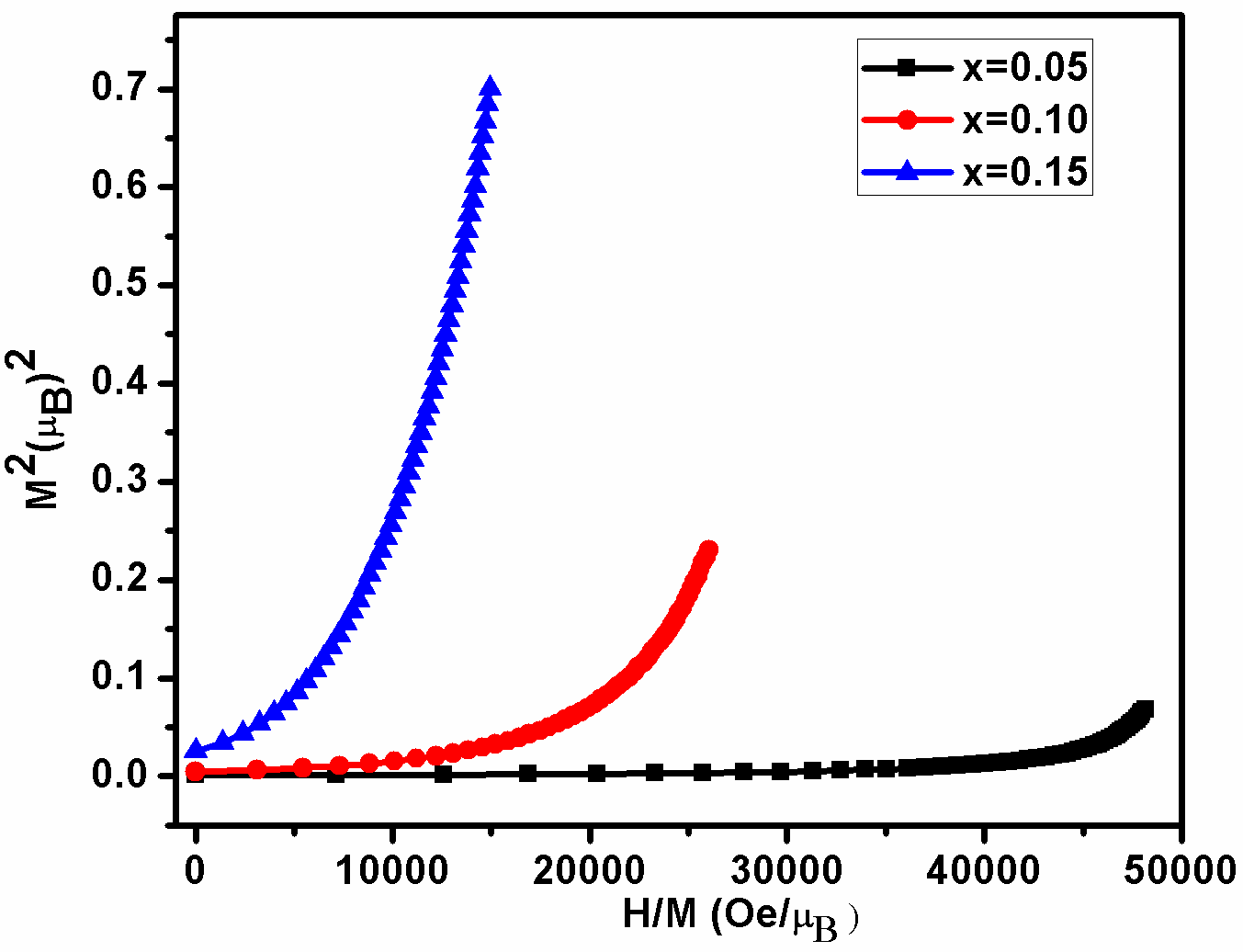
BDF1, BDF2 and BDF3 nanoparticles, we have carried out a magnetic field-dependent magnetization (M-H) and the magnetic field range from 0 to ±15 kOe at room temperature published elsewhere.[29] Furthermore confirmation of magnetic properties, we have evaluated from magnetic measurement (H/M) - as a function of (M2) plotted in Arrott–Belov–Kouvel (ABK) model which are shown in figure 8 at room temperature for BDF1, BDF2 and BDF3. In the ABK plots for the BDF3 sample exhibit a concave behavior without any spontaneous magnetization, indicating an antiferromagnetism.[43] However, BDF2 and BDF3 the marked convex nature of each isothermal curve with spontaneous magnetization (having positive intercept in the M2 axis at H=0) corroborates the presence of the ferromagnetism behavior.[44]
3.9. Magneto-Electric Effect Analysis
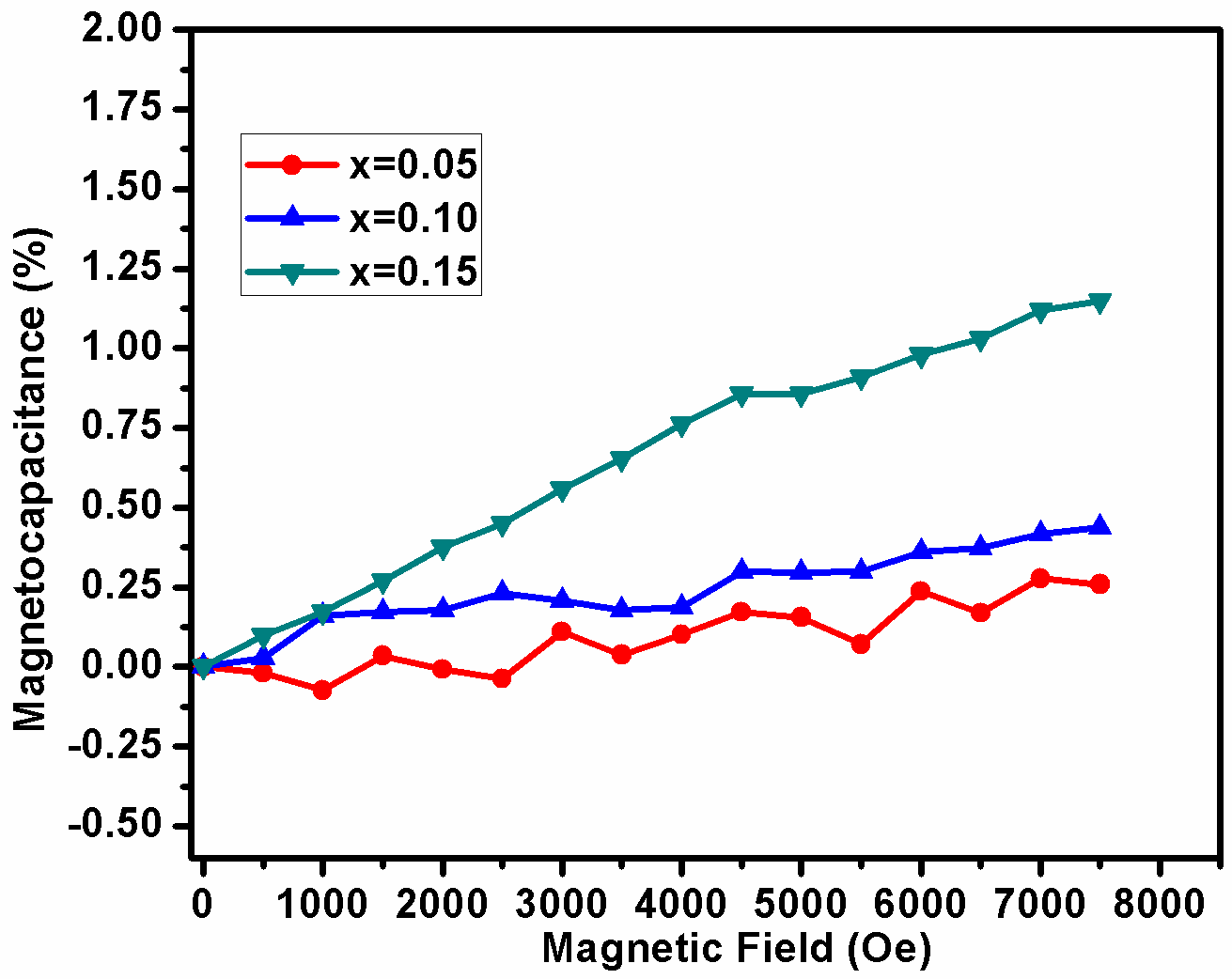
In order to demonstrate the magneto electric coupling of BDF1, BDF2 and BDF3 samples, measured capacitance with applied magnetic field at room temperature as shown in Fig.9. It has been observed that the value of magneto-capacitance is increases with increasing Dy concentration into BFO. Similar phenomena have also been observed in rare earth doped BFO ceramics.[45,46] From these results, one can predict the existence of magnetoelectric coupling in the system which increases with increase in Dy concentration. We expected these results will be useful for application towards development of current probe, advanced logic devices, magnetic field sensing and ME energy harvester.
4. Conclusions
BDF1, BDF2 and BDF3 nanoparticles successfully synthesised by co-precipitation method at low temperature. Structural transformation from rhombohedral to orthorhombic structure with increase in the Dy concentration into BFO. The average crystallite sizes and micro strain were calculated from W-H plot. The formation of perovskite structure can be confirmed by the presence of metal-oxygen band. The band gap energy found to be 2.18, 2.16 and 2.13 eV for BDF1, BDF2 and BDF3 samples respectively. Four emission peaks positioned at 411 (3.01 eV), 437 (2.83 eV), 468 (2.64 eV) and 539 nm (2.30 eV) have been observed in photoluminescence. From thermal analysis observed the two distinct peaks as ferroelectric ordering and melting point temperature. The leakage current density was found to be reduced with increasing Dy concentration in the order of 10−7 A/cm2 at 90 V/cm. To confirm the antiferromagnetic behavior of BDF1 and also BDF2 and BDF3 observed ferromagnetic behavior at room temperature from ABK plots are calculated from M–H data. Further, we have demonstrated the magneto electric effect for the BDF1, BDF2 and BDF3 samples.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Prellier W.; Singh M. P.; Murugavel P. The Single-Phase Multiferroic Oxides: from Bulk to Thin Film. J. Phys. Condens. Matter., 2005, 17, R803–R832. [CrossRef]
- Catalan C.; Scott J. F. Physics and Applications of Bismuth Ferrite. Advanced Materials., 2009, 21, 2463–2485. [CrossRef]
- Smolenskiy G.A.; Chupis I. E. Ferroelectromagnets. Soviet Physics Uspekhi., 1982, 25, 475. [CrossRef]
- Sosnowska.; Peterlin-Neumaier T and Steichele E. Spiral Magnetic Ordering in Bismuth Ferrite. J. Phys. C: Solid State Phys., 1982, 15, 4835. [CrossRef]
- Cheng Z. X.; Li A. H.; Wang X. L.; Dou S. X.; Ozawa K.; Kimura H.; Zhang S. J. and Shrout T. R. Structure, Ferroelectric Properties Magnetic Properties of the La-doped Bismuth Ferrite. J. Appl. Phys., 2008, 10, 307-507. [CrossRef]
- Wang J. B. N. J.; Neaton J. B.; Zheng H.; Nagarajan V.; Ogale S. B.; Liu B.; Viehland D.; Vaithyanathan V.; Schlom D. G.; Waghmar U. V.; Spaldin N. A. Epitaxial BiFeO3 Multiferroic Thin Film Heterostructures. Science, 2003, 299, 1719–1722. [CrossRef]
- Jeon N.; Rout D.; Kim I. W.; Kang S. J. L. Enhanced Multiferroic Properties of Single-Phase BiFeO3 Bulk Ceramics by Ho Doping. Appl. Phys. Lett., 2011, 98, 072901. [CrossRef]
- Muneeswaran M.; Dhanalakshmi R.; Giridharan N. V. Structural, Vibrational, Electrical and Magnetic Properties of Bi1−xPrxFeO3. Ceram. Int., 2015, 41, 8511–8519. [CrossRef]
- Muneeswaran M.; Dhanalakshmi R.; Giridharan N. V. Effect of Tb Substitution on Structural, Optical, Electrical and Magnetic Properties of BiFeO3. J Mater Sci: Mater Electron, 2015, 26, 3827–3839. [CrossRef]
- Chauhan S.; Kumar M.; Chhoker S.; Katyal S. C.; Singh H.; Jewariya M.; Yadav K. L. Multiferroic, Magnetoelectric and Optical Properties of Mn Doped BiFeO3 Nanoparticles. Solid State Commun., 2012, 152, 525–529. [CrossRef]
- Sui Y.; Xin C.; Zhang X.; Yang Y.; Wang Y.; Wang X.; Liu Z.; Li B.; Liu X. Enhancement of Multiferroic in BiFeO3 by Co Doping. J. Alloys Compd., 2015, 645, 78–84. [CrossRef]
- Bernardo M. S.; Jardial T.; Peiteado M.; Garcia F. J.; Villegas M.; Cabaluro A. C. Intrinsic Compositional Inhomogeneities in Bulk Ti-Doped BiFeO3: Microstructure Development and Multiferroic Properties. Chem. Mater., 2013, 25, 1533–1541. [CrossRef]
- Mao W.; Yao Q.; Fan Y.; Wang Y.; Wang X.; Pu Y.; Li X. Combined Experimental and Theoretical Investigation on Modulation of Multiferroic Properties in BiFeO3 Ceramics Induced by Dy and Transition Metals Co-Doping. J. Alloys Compd., 2019, 784, 117-124. [CrossRef]
- Gumiel C.; Jardiel T.; Bernardo M. S.; Villanueva P. G.; Urdirozc U.; Cebollada F.; Aragód C.; Caballero A. C.; Peiteado M. Combination of Structural and Microstructural Effects in the Multiferroic Response of Nd and Ti Co-Doped BiFeO3 Bulk Ceramics. Ceram. Inter., 2019, 45, 5276–5283. [CrossRef]
- Anwar A.; Basith M.A.; Choudhury S. From Bulk to Nano: A Comparative Investigation of Structural, Ferroelectric and Magnetic Properties of Sm and Ti Co-Doped BiFeO3 Multiferroics. Mater. Res. Bull., 2019, 111, 93–101. [CrossRef]
- Fruth V.; Tenea E.; Gartner M.; Anastasescu M.; Berger D.; Ramer R.; Zaharescu M. Preparation of BiFeO3 Films by Wet Chemical Method and Their Characterization. J. Eur. Ceram Soc., 2007, 27, 937–940. [CrossRef]
- Sati P. C.; Arora M.; Chauhan S.; Kumar M.; Chhoker S. Effect of Dy Substitution on Structural, Magnetic and Optical Properties of BiFeO3 Ceramics. J Phys Chem Solids, 2014, 75,105–108. [CrossRef]
- Peng L.; Deng H.; Tian J.; Ren Q.; Peng C.; Huang Z.; Yang P.; Chu J. Influence of Co Doping on Structural, Optical and Magnetic Properties of BiFeO3 Films Deposited on Quartz Substrates by Sol–Gel Method. Appl. Surf. Sci., 2013, 268, 146– 150. [CrossRef]
- Gu B.; Wang Y.; Wang J.; Ji W. Femtosecond Third-Order Optical Nonlinearity of Polycrystalline BiFeO3. Optics Express., 2009, 17, 10970. [CrossRef]
- Gao F.; Chen X.; Yin K.; Dong S.; Ren Z.; Yuan F.; Yu T.; Zou Z.; Liu J. M. Visibleâ€Light Photocatalytic Properties of Weak Magnetic BiFeO3 Nanoparticles. Adv. Mater., 2007, 19, 2889. [CrossRef]
- Choi T.; Lee S.; Choi Y. J.; Kiryukhin V.; Cheong S. W. Switchable Ferroelectric Diode and Photovoltaic Effect in BiFeO3. Science, 2009, 324, 63. [CrossRef]
- Lotey G. S.; Verma N. K. Phase-Dependent Multiferroism in Dy-Doped BiFeO3 Nanowires. Superlattices and Microstructures, 2013, 53, 184–194. [CrossRef]
- Chowdhury S. S.; Kamal A. H. M.; Hossain R.; Hasan M.; Islam Md. F.; Ahmmad B.; Basith M. A. Dy Doped BiFeO3: A Bulk Ceramic with Improved Multiferroic Properties Compared to Nano Counterparts. Ceram. Inter., 2017, 43, 9191–9199. [CrossRef]
- Koval V.; Skorvanek I.; Reece M.; Mitoseriu L.; Yan H. Effect of Dysprosium Substitution on Crystal Structure and Physical Properties of Multiferroic BiFeO3 Ceramics. J. Eur. Ceram. Soc., 2014, 34, 641-651. [CrossRef]
- Khomchenko V. A.; Karpinsky D. V.; Kholkin A. L.; Sobolev N. A. and Kakazei G. N. Rhombohedral-to-Orthorhombic Transition and Multiferroic Properties of Dy-Substituted BiFeO3. J. Appl. Phys., 2010, 108, 074109. [CrossRef]
- Muneeswaran M. and Giridharan N. V. Effect of Dy-Substitution on the Structural, Vibrational, and Multiferroic Properties of BiFeO3 Nanoparticles. J. Appl. Phys., 2014, 115, 214109. [CrossRef]
- Xu J.; Ye G.; Zeng M. Structure Transition and Enhanced Multiferroic Properties of Dy-Doped BiFeO3. J. Alloy. Compd., 2014, 587, 308-312. [CrossRef]
- Sun C.; Chen X.; Wang J.; Yuan G.; Yin J.; Liu Z. Structure and Piezoelectric Properties of BiFeO3 and Bi0.92Dy0.08FeO3 Multiferroics at High Temperature. Solid State Commun., 2012, 152, 1194-1198. [CrossRef]
- Muneeswaran M.; Jegatheesan P.; Gopiraman M.; Kim I. S. and Giridharan N. V. Structural, Optical, and Multiferroic Properties of Single Phased BiFeO3. Appl. Phys. A., 2014, 114, 853–859. [CrossRef]
- Muneeswaran M.; Jegatheesan P.; Giridharan N. V. Synthesis of Nanosized BiFeO3 Powders by Co-Precipitation Method. J. Exp. Nanosci., 2013, 8, 341–346. [CrossRef]
- Marezio M.; Remeika J. P. and Dernier P. D. The Crystal Chemistry of the Rare Earth Orthoferrites. Acta Crystallogr., Sect. B: Struct. Crystallogr. Cryst. Chem., 1970, 26, 2008. [CrossRef]
- Streltsov V. A. and Ishizawa N. Synchrotron X-ray Study of the Electron Density in RFeO3 (R = Nd, Dy). Acta Crystallogr., Sect. B: Struct. Sci., 1999, 55, 1. [CrossRef]
- Chen Z.; Huang C.; Qi Y.; Yang P.; You L.; Hu C.; Wu T.; Wang J.; Gao C.; Sritharan T.; Chen L. Lowâ€Symmetry Monoclinic Phases and Polarization Rotation Path Mediated by Epitaxial Strain in Multiferroic BiFeO3 Thin Films. Adv. Funct. Mater., 2011, 21, 133–138. [CrossRef]
- Mokkelbost T.; Kaus I.; Grande T.; Einarsrud M. A. Combustion Synthesis and Characterization of Nanocrystalline CeO2-Based Powders. Chem. Mater., 2004, 16, 5489. [CrossRef]
- Nowak M.; Kauch B. and Szperlich P. Determination of Energy Band Gap of Nanocrystalline SbSI Using Diffuse Reflectance Spectroscopy. Rev. sci. inst., 2009, 80, 046107. [CrossRef]
- Ramachandran B.; Dixit A.; Naik R.; Lawes G.; Rao M. S. R. Charge Transfer and Electronic Transitions in Polycrystalline BiFeO3. Phys. Rev. B. 2010, 82, 012102. [CrossRef]
- Elilarassi R.; Chandrasekaran G. Structural, Optical and Magnetic Properties of Nanoparticles of ZnO:Ni—DMS Prepared by Sol–Gel Method. Mater. Chem. Phys., 2010, 123, 450-455. [CrossRef]
- Soram B. S.; Ngangom B. S.; Sharma H. B. Effect of Annealing Temperatures on the Structural and Optical Properties of Sol–Gel Processed Nanocrystalline BiFeO3 Thin Films. Thin Solid Films., 2012, 524, 57–61. [CrossRef]
- Das S. R.; Choudhary R. N. P.; Bhattacharya P. and Katiyar R. S.; Dutta P.; Manivannan A. and Seehra M. S. Structural and Multiferroic Properties of La-Modified BiFeO3 Ceramics. J. Appl. Phys., 2007, 101, 034104. [CrossRef]
- Singh S. K.; Maruyama K.; Ishiwara H. Reduced Leakage Current in La and Ni Co-Doped BiFeO3 Thin Films. Appl. Phys. Lett., 2007, 91, 112913. [CrossRef]
- Prashanthi K.; Chalke B. A; Barick K. C.; Das A.; Dhiman I.; Palkar V. R. Enhancement in multiferroic properties of Bi0.7-xLaxDy0.3FeO3 system with removal of La. Solid State Communications, 2009, 149, 188-191. [CrossRef]
- Zhang S.; Wang L.; Chen Y.; Wang D.; Yao Y.; Ma Y. Observation of Room Temperature Saturated Ferroelectric Polarization in Dy Substituted BiFeO3 Ceramics. J. Appl. Phys., 2012, 111, 074105. [CrossRef]
- Nath T. K.; Sudhakar N.; McNiff E. J.; Majumdar A. K. Magnetization Study of γ-Fe80−xNixCr20 (14⩽x⩽30) Alloys to 20 T. Phys. Rev. B. 1997, 55, 12389. [CrossRef]
- Singh V.; Srinivas V.; Ranot M.; Angappane S.; Geun Park J. Effect of Polymer Coating on the Magnetic Properties of Oxygen-Stabilized Nickel Nanoparticles. Phys. Rev. B., 2010, 82, 054417. [CrossRef]
- Muneeswaran M.; Lee S.H.; Kim D. H.; Jung B. S.; Chang S. H.; Jang m J. W.; Choi B. C.; Jeong J. H.; Giridharan N. V.; Venkateswaran C. Structural, Vibrational, and Enhanced Magneto-Electric Coupling in Ho-Substituted BiFeO3. J. Alloy. Compd., 2018, 750, 276-285. [CrossRef]
- Ruette B.; Zvyagin S.; Pyatakov A.P.; Bush A.; Li J. F.; Belotelov V. I.; Zvezdin A. K.; Viehland D. Magnetic-Field-Induced Phase Transition in BiFeO3 Observed by High-Field Electron Spin Resonance:Cycloidal to Homogeneous Spin Order. Phys. Rev. B., 2004, 69, 064114. [CrossRef]
 © 2019, by the authors. Licensee Ariviyal Publishing, India. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
© 2019, by the authors. Licensee Ariviyal Publishing, India. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).


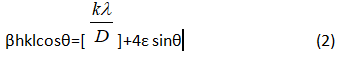
.PNG)
.PNG)