Chemical Science & Engineering Research
Title
Infrared (Attenuated Total Reflection) Study of Lithium Salts Dissolved in Non-Aqueous Aprotic Solvents
Authors
Sharda Sundaram Sanjay,*a Mrigank Mauli Dwivedib and Kamlesh Pandeyb
aDepartment of Chemistry, Ewing Christian College, University of Allahabad, Allahabad-211002, U.P., India.
bNational Center of Experimental Mineralogy and Petrology, University of Allahabad, Allahabad-211002, U.P., India.
*Corresponding author E-mail address: sharda100@rediffmail.com (Sharda Sundaram Sanjay)
Article History
Publication details: Received: 04th November 2022; Revised: 17th November 2022; Accepted: 17th November 2022; Published: 03rd December 2022
Cite this article
Sharda S.S.; Dwivedi M.M.; Pandey K. Infrared (Attenuated Total Reflection) Study of Lithium Salts Dissolved in Non-Aqueous Aprotic Solvents. Chem. Sci. Eng. Res., 2022, 4(11), 43-47.
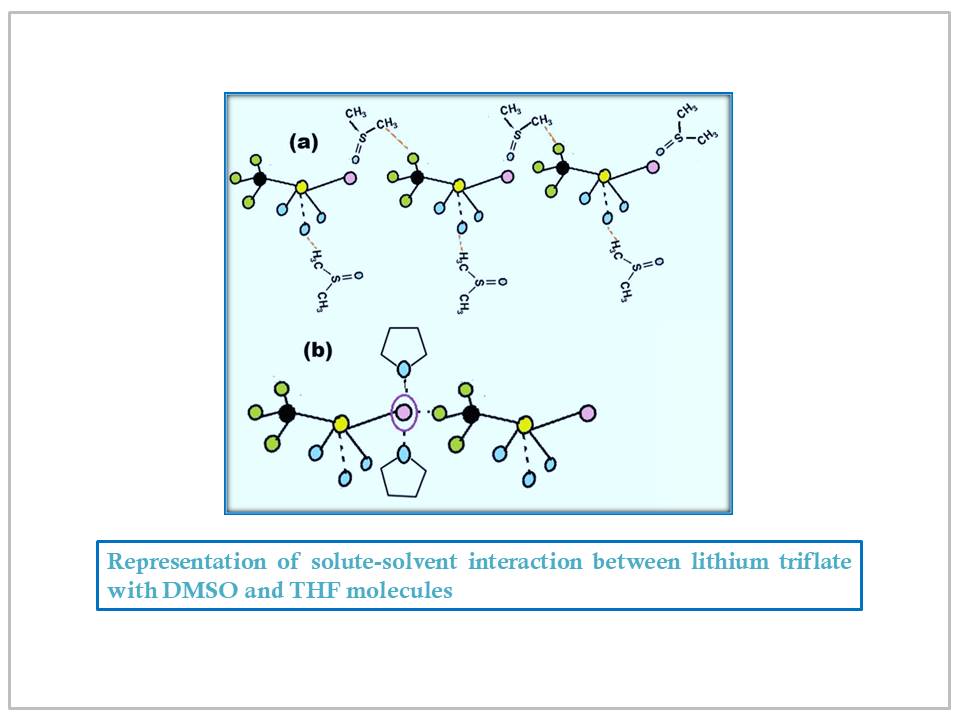
Abstract
The role of the solvent becomes critically important during ion transport when the electrolyte solute and solvent compete for coordination. Improved ionic conductivity requires ionic dissociation. The 'hip-hop' transition of the cations requires that ion pairs split and become solvated with solvent molecules during dissolution. To achieve these hopping transitions, lithium salts are the best choice. We have investigated the vibrational spectra of lithium nitrate and lithium triflate in two different non-aqueous organic solvents, namely THF and DMSO, through ATR-FTIR spectroscopy in order to investigate ion-solvent and ion-ion interactions, to learn more about solvents' lower and higher frequency vibrations, and factors affecting the identifiable characteristics, functioning, and interaction of ionic compounds with solvents.
Keywords
FTIR; Lithium Nitrate; Lithium Triflate; THF; DMSO; Solute-Solvent Interaction